2007 Spring Symposium
Al Metauro
Hyperion Catalysis International Inc.
Cambridge, MA
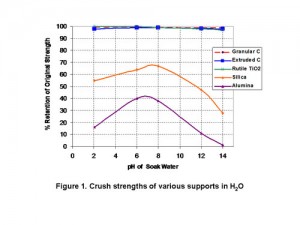
Abstract — Forms of carbon have been known and used commercially as supports for active metal (and compounds of metal) catalysts for many years. It has been found that the distinctive properties of multi-wall carbon nanotube aggregates can be utilized to prepare durable supports for slurry and fixed bed catalytic applications that show enhanced activity in selected reactions. The activity benefits from the high level of porosity in the mesopore range found in the support structure and the fact that the active catalytic metals can be dispersed efficiently throughout the carbon nanotube support. In addition the supports show increased attrition resistance and strength.
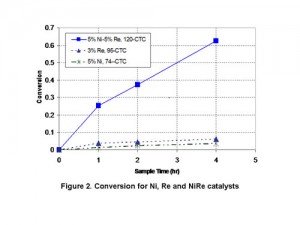
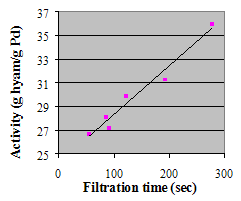
The presentation will discuss several applications for carbon nanotube supports found in the literature. Data on physical properties and specific test results for the hydrogenation of cyclohexene to cyclohexane and the hydrogenation of nitrobenzene to aniline in comparison with activated carbon catalysts will be shown. Hyperion Catalysis is the leading producer of multi-wall carbon nanotubes and can supply consistent commercial products that can add value to many chemical processes.